Molecular orbital molecule orbitals structures unizin wisc
Table of Contents
Table of Contents
Are you struggling with how to draw molecular orbital energy level diagrams? Look no further, as this article will provide you with a step-by-step guide to drawing these diagrams successfully.
Many students find it challenging to draw molecular orbital energy level diagrams due to the complex nature of the topic. It can be challenging to visualize how the orbitals overlap and how electrons fill them. Additionally, it can seem overwhelming to understand the variables that contribute to the energy levels of orbitals.
Drawing a molecular orbital energy level diagram involves understanding the energy levels of atomic orbitals, identifying the number of electrons in each orbital, and drawing energy level diagrams for both the atomic and molecular orbitals.
In summary, drawing molecular orbital energy level diagrams involves analyzing the energy levels of atomic orbitals, understanding the number of electrons in each orbital, and drawing orbitals for both atomic and molecular levels.
How to Draw Molecular Orbital Energy Level Diagrams: A Step-by-Step Guide
When drawing molecular orbital energy level diagrams, there are a few essential steps you should follow:
First, identify the atomic orbitals of each atom in the molecule.
Next, analyze the number of electrons in each atomic orbital.
Then, draw the molecular orbitals, taking into account the number of electrons in each molecular orbital and their relative energies.
Finally, fill the molecular orbitals with electrons, following the Pauli Exclusion Principle and Hund’s Rule.
Here is a more detailed explanation of each step:
Step 1: Identify the Atomic Orbitals of Each Atom in the Molecule
Before drawing a molecular orbital energy level diagram, you must identify the atomic orbitals of each atom in the molecule. The atomic orbitals include the s, p, d, and f orbitals.
For example, suppose you want to draw the molecular orbital energy level diagram for the molecule H2, which contains two hydrogen atoms. In that case, you would identify the atomic orbitals of hydrogen, which include the 1s orbital.
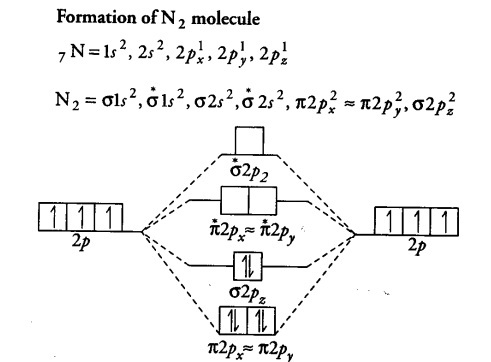 ### Step 2: Analyze the Number of Electrons in Each Atomic Orbital
### Step 2: Analyze the Number of Electrons in Each Atomic Orbital
Once you have identified the atomic orbitals, you must analyze the number of electrons in each atomic orbital. This information is typically written in the electron configuration of each atom.
For example, each hydrogen atom has one electron in the 1s orbital. Therefore, the molecule’s two hydrogen atoms have a total of two electrons in the 1s orbital.
Step 3: Draw the Molecular Orbitals
After analyzing the number of electrons in each atomic orbital, you can draw the molecular orbitals. Molecular orbitals describe the probability of finding electrons in a particular region between two atoms.
When drawing the molecular orbitals, you must consider the atomic orbitals’ energy levels and how they will overlap with each other.
Step 4: Fill the Molecular Orbitals with Electrons
After drawing the molecular orbitals, you must fill them with electrons by following the Pauli Exclusion Principle and Hund’s Rule. The Pauli Exclusion Principle states that no two electrons can have the same set of four quantum numbers. Hund’s Rule states that, when possible, electrons will occupy an empty orbital before pairing up in the same orbital.
What are the Benefits of Drawing Molecular Orbital Energy Level Diagrams?
There are several benefits to drawing molecular orbital energy level diagrams. One benefit is that they provide insight into how electrons are distributed among the different energy levels in a molecule.
They also help to explain the strength and stability of chemical bonds. By understanding the molecular orbitals and their energies, you can determine how the atoms in the molecule interact and how easy it is to break or form bonds.
Common Mistakes to Avoid When Drawing Molecular Orbital Energy Level Diagrams
Here are a few common mistakes to avoid when drawing molecular orbital energy level diagrams:
Mistake 1: Neglecting to Consider the Relative Energies of Orbitals
When drawing molecular orbital energy level diagrams, it is essential to consider the relative energy levels of the orbitals. Neglecting to do so can lead to incorrect diagrams.
Mistake 2: Not Following the Pauli Exclusion Principle and Hund’s Rule
Pauli Exclusion Principle and Hund’s Rule must be followed when filling molecular orbitals with electrons. Neglecting to follow these rules can lead to incorrect diagrams and a misunderstanding of the molecule’s electronic structure.
Question and Answer
Q: What is a molecular orbital energy level diagram?
A: A molecular orbital energy level diagram is a graphical representation of the relative energies and electron distributions of molecular orbitals in a molecule.
Q: Why are molecular orbital energy level diagrams important?
A: Molecular orbital energy level diagrams help to explain the strength and stability of chemical bonds and provide insight into how electrons are distributed among different energy levels in a molecule.
Q: What is the difference between an atomic orbital and a molecular orbital?
A: An atomic orbital describes the probability of finding an electron in a particular region around an atom, while a molecular orbital describes the probability of finding electrons in a particular region between two atoms.
Q: What is the Pauli Exclusion Principle?
A: The Pauli Exclusion Principle states that no two electrons in an atom can have the same set of four quantum numbers.
Conclusion of How to Draw Molecular Orbital Energy Level Diagrams
Drawing molecular orbital energy level diagrams can seem challenging at first, but by following the steps outlined in this article, you can create accurate diagrams confidently. Understanding the relative energies and electron distributions of molecular orbitals can provide insight into the strength and stability of chemical bonds and the electron distribution in a molecule. Always remember to follow Pauli Exclusion Principle and Hund’s Rule when filling molecular orbitals with electrons, and consider the relative energy levels of orbitals when drawing your diagrams.
Gallery
9.8: Second-Row Diatomic Molecules - Chemistry LibreTexts
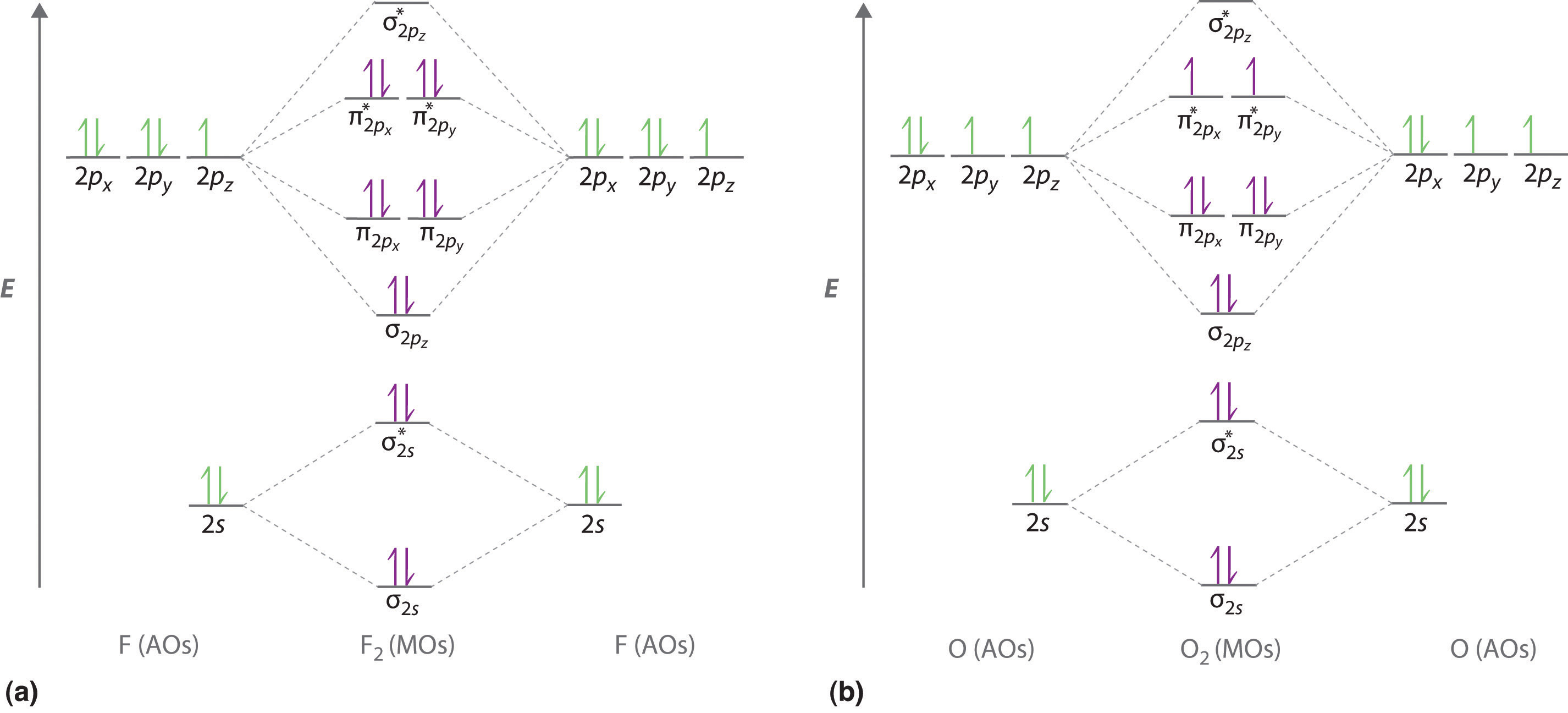
Photo Credit by: bing.com / orbital nh3 orbitals diatomic molecules of2 bonding delocalized libretexts chem homonuclear o2 valence electrons techiescientist hybridization pageindex conclusion
Day 6: Molecular Orbitals; Lewis Structures – Chemistry 109, Fall 2020
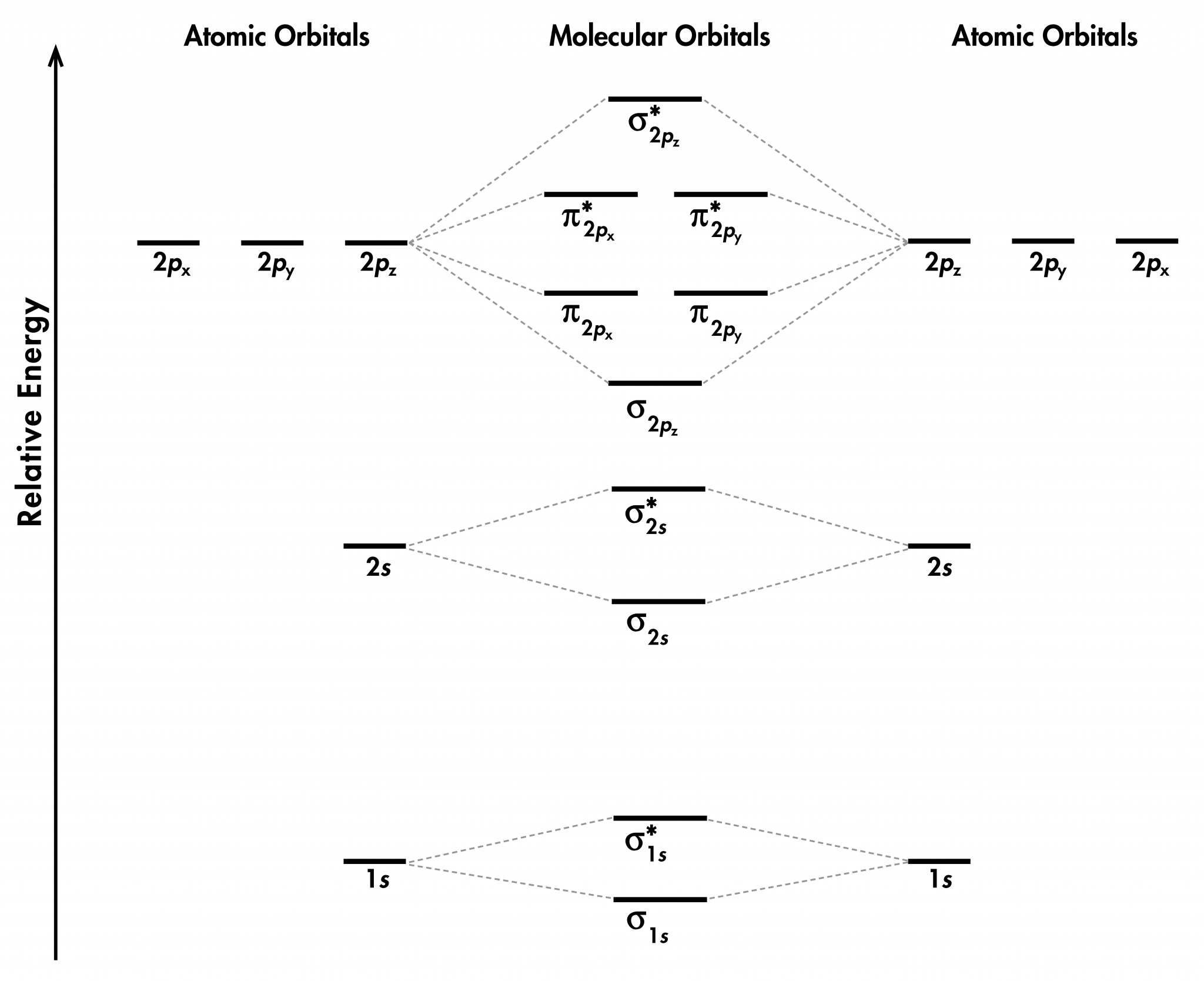
Photo Credit by: bing.com / molecular orbital molecule orbitals structures unizin wisc
Solved: 1.a) Draw The Molecular Orbital Energy Level Schem… | Chegg.com
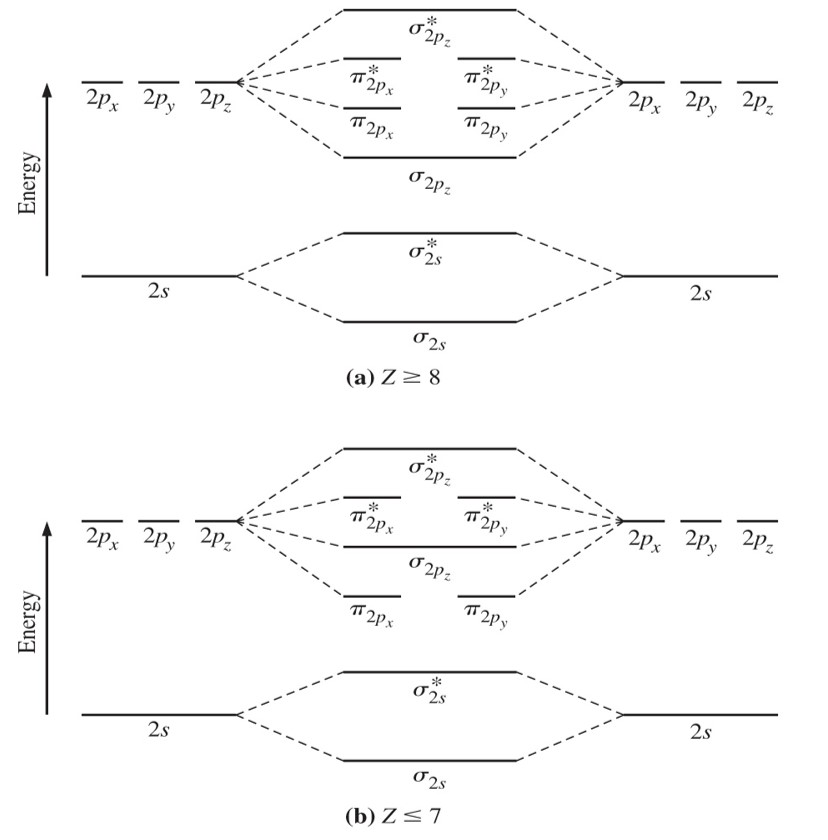
Photo Credit by: bing.com / energy level orbital molecular draw mo diagram bond schem solved period chegg order predict drawing use second give
Energy Level Diagram For Molecular Orbitals - Chemical Bonding And
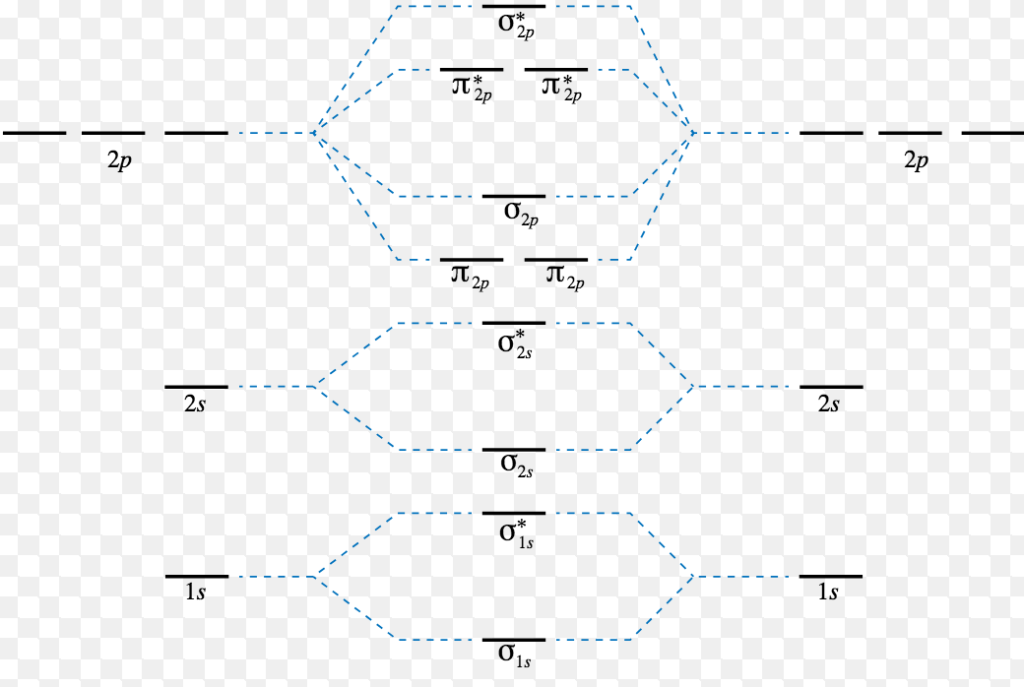
Photo Credit by: bing.com / energy molecular level diagram orbitals orbital n2 o2 f2 bonding structure order chemistry ne2
Use The Molecular Orbital Energy Level Diagram To Show That - CBSE

Photo Credit by: bing.com / diagram orbital molecular energy level show class chemistry use prasanna 55am december





